Keywords
Abstract
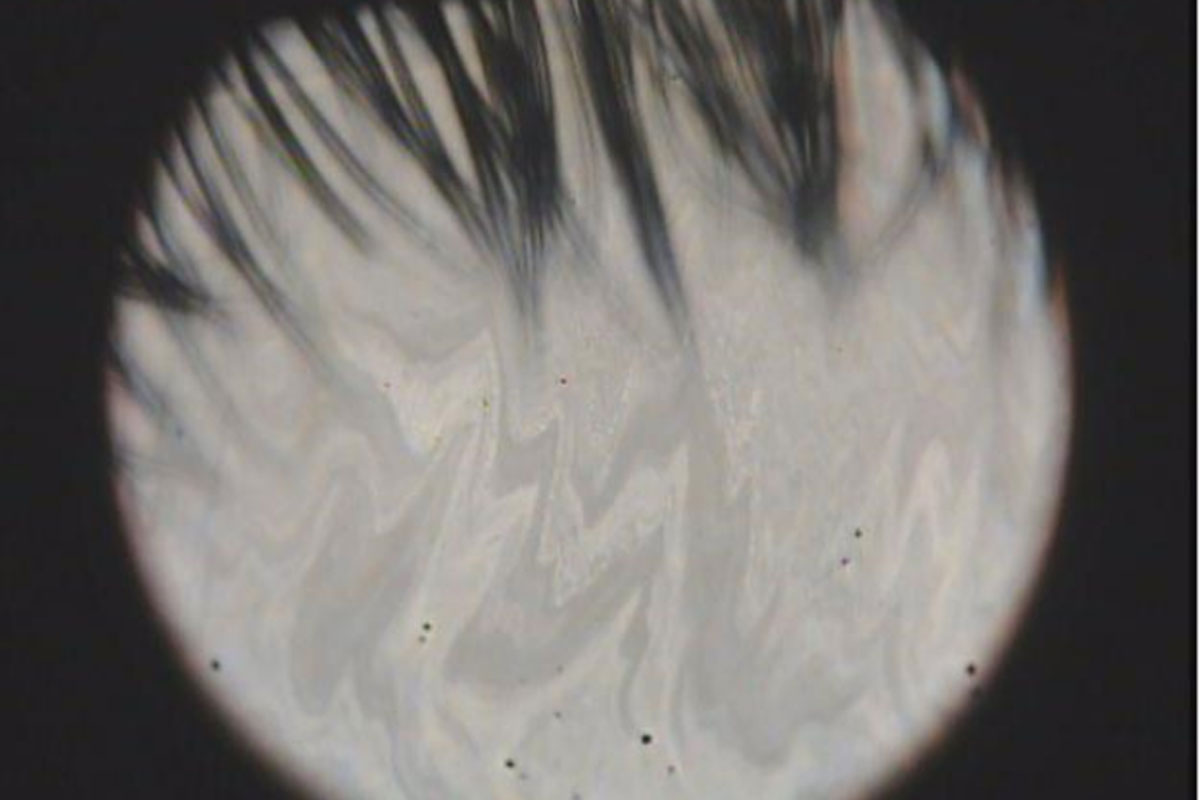
The unstable balance in secretions of lipids and aqueous fluid to tear film is a significant cause of dry eye disease (DED). Arita et al. demonstrated a simple but very effective method that classifies dry eye types to the aqueous deficient dry eye (ADDE) and the evaporative dry eye (EDE) by focusing on the dry eye type-unique appearances of interference fringe colors and patterns of tear films. We thought this simple classification is very helpful for diagnoses and treatments. However, diagnostic bias by unskilled observers remains an issue to be solved.
The artificial intelligence (AI)-based support for diagnosis is one of the hottest topics in the field of ophthalmology research. We expected that the AI-based model would reduce bias in DED-type diagnoses. Many studies have been reported targeting retinal diseases like age-related macular degeneration and/or diabetic retinopathy. Most of the works established AI-based predicting models using images taken by fundus cameras and/or optical coherence tomography (OCT) devices to capture disease-related structural disorders. In contrast, the interference fringes dynamically change the colors and patterns spatiotemporally. To the best of our knowledge, there is no AI-based model studied for distinguishing ADDE and EDE using interference fringe images. However, an AI-based study classifying the condition of the tear lipid layer by analyzing the textures of interference fringes compared to the device-unique grades has been reported. This suggested the possibility of using the unstructured characteristics, such as colors and/or complexities of interference fringes, as the numerical image features when building AI-based prediction models. In this study, we first examined several types of image characteristics extracted from the colors and patterns of fringes to obtain effective image features for the DED-type classification. We then evaluated whether the AI-based models would have sufficient abilities for this type of prediction by comparing their diagnoses with those made by an ophthalmologist skilled in this classification (the founder of this type classification).
References
Craig JP, Nichols KK, Akpek EK, et al. TFOS DEWS II Definition and Classification Report. Ocul Surf. 2017;15: 276-283.
Arita R, Morishige N, Koh S, et al. Increased tear fluid production as a compensatory response to Meibomian gland loss: a multicenter cross-sectional study. Ophthalmology. 2015;122: 925-933.
Arita R, Morishige N, Fujii T, et al. Tear interferometric patterns reflect clinical tear dynamics in dry eye patients. IOVS. 2016;57: 3928-3934.
Liu YY, Chen M, Ishikawa H, Wollstein G, Schuman JS, Rehg JM. Automated macular pathology diagnosis in retinal OCT images using multi-scale spatial pyramid and local binary patterns in texture and shape encoding. Med Image Anal. 2011;15: 748-759.
Schmidt-Erfurth U, Klimscha S, Waldstein SM, Bogunović H. A view of the current and future role of optical coherence tomography in the management of age-related macular degeneration. Eye. 2017;31: 26-44.
Ataer-Cansizoglu E, Bolon-Canedo V, Campbell JP, et al. Computer-based image analysis for plus disease diagnosis in retinopathy of prematurity: performance of the “i-ROP” system and image features associated with expert. TVST. 2015;4: Article 5.
Jaya T, Dheeba J, Singh NA. Detection of hard exudates in colour fundus images using fuzzy support vector machine-based expert system. J Digit Imaging. 2015;28: 761-768.
Remeseiro B, Bolon-Canedo V, Peteiro-Barral D, et al. A methodology for improving tear film lipid layer classification. IEEE J Biomed Health Inform. 2014;18: 1485-1493.